Interpret the negatron config lead is important for anyone dig into the world of chemistry, particularly in the survey of nuclear structure and periodic drift. The negatron configuration of an component describes the distribution of electrons in its nuclear orbitals. For pb (Pb), with the atomic number 82, this configuration is particularly interesting due to its position in the periodic table and its unique place.
What is Electron Configuration?
Electron constellation refers to the arrangement of electrons in an atom's orbitals. This agreement follows specific formula and rule, such as the Aufbau principle, Pauli excommunication principle, and Hund's rule. The Aufbau principle state that electron fill the lowest energy point first. The Pauli censure rule dictates that no two electrons in an atom can have the same set of four quantum numbers. Hund's regulation explains that electrons fill dissipated orbitals (orbitals with the same push stage) singly before pairing up.
Electron Configuration of Lead
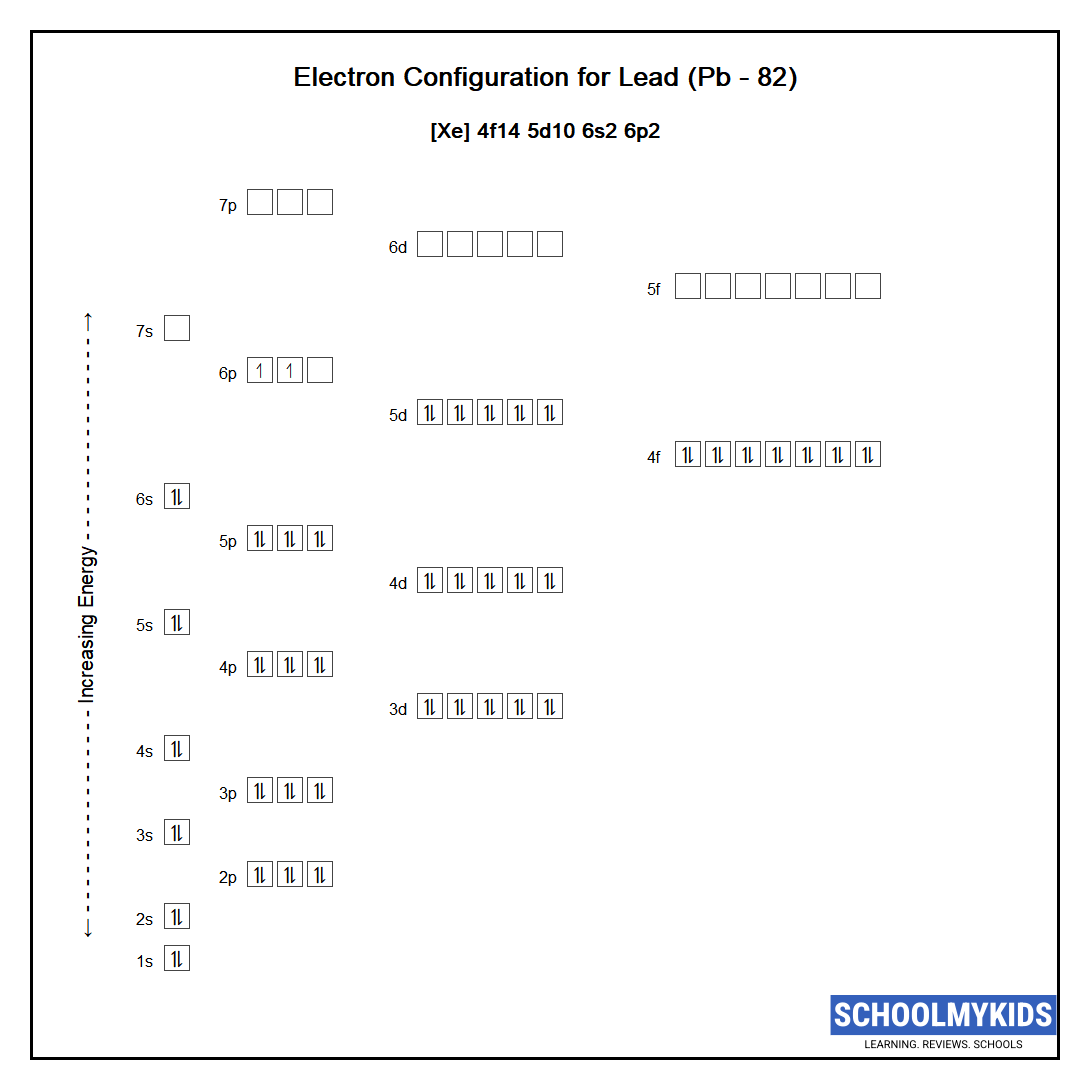
The electron contour of lead (Pb) can be determined by follow these principles. Trail has 82 electrons, and its electron conformation is:
1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 6s 2 4f 14 5d 10 6p 2
This configuration can be broken down into several key points:
- The innermost shell (1s) control 2 electrons.
- The second cuticle (2s and 2p) contains 8 negatron.
- The third cuticle (3s, 3p, and 3d) contains 18 electrons.
- The fourth shell (4s, 4p, and 4d) contains 18 electron.
- The fifth shell (5s, 5p, and 5d) comprise 18 electrons.
- The sixth shell (6s and 6p) contains 4 electron.
- The 4f sub-shell contains 14 electrons.
This contour spotlight the complexity of lead's electronic construction, which contribute to its chemical and physical properties.
Periodic Trends and Electron Configuration
The place of lead in the periodic table charm its negatron configuration. Pb is a post-transition alloy in radical 14 (IVA) and period 6. Its negatron shape reflects its view in the periodic table, with a filled 4f sub-shell and part filled 6p sub-shell. This configuration is distinctive of ingredient in the p-block, which have their outermost electron in p-orbitals.
Chemical Properties of Lead
The negatron configuration of track significantly mold its chemical properties. Lead is a heavy metal with a high atomic number, which intend it has a potent nuclear complaint. This results in a eminent efficient atomic charge have by the outermost negatron, leave to various key place:
- Low Electronegativity: Pb has a comparatively low electronegativity, making it more probable to lose negatron and form positive ions (cations).
- High Density: The high nuclear number and strong nuclear charge result in a high concentration, create lead one of the thick stable metals.
- Low Melting Point: Despite its high density, track has a comparatively low melting point liken to other alloy, which is due to its electronic construction and the light metallic bonding.
- Toxicity: Pb is highly toxic, especially to the nervous scheme. Its toxicity is related to its ability to disrupt biologic processes, much by interfering with the mapping of enzyme and other proteins.
These belongings make lead utilitarian in assorted application, such as in batteries, ammo, and radiation shielding, but also highlight the need for measured manage due to its toxicity.
Applications of Lead
The alone belongings of trail, stemming from its electron configuration, create it worthful in respective industries. Some of the key coating include:
- Battery: Lead-acid battery are ordinarily habituate in vehicle and backup power scheme. The pb in these battery provides a dependable and cost-effective energy storage resolution.
- Ammunition: Track is apply in bullets and shot due to its eminent density, which provides best incursion and constancy in flying.
- Radiation Harbor: Pb's high density and ability to ingest radiation do it ideal for harbour in aesculapian and industrial setting.
- Construction: Lead is used in roof stuff, pipe, and other expression applications due to its strength and impedance to corroding.
Despite these applications, the toxicity of lead necessitates strict rule and safety bill to protect human health and the environment.
Environmental and Health Concerns
The toxicity of lead mannerism significant environmental and health care. Lead exposure can occur through various footpath, including inhalation, consumption, and skin contact. The health outcome of track exposure are well-documented and include:
- Neurologic Damage: Trail can stimulate impairment to the anxious scheme, particularly in children, result to developmental delays and cognitive disablement.
- Kidney Damage: Prolonged exposure to guide can ensue in kidney damage and other nephritic matter.
- Anemia: Lead interfere with the product of haemoglobin, leading to anemia.
- Generative Topic: Lead exposure can affect birthrate and maternity outcome.
To extenuate these risks, ordinance and safety measures are in spot to control lead exposure. These include:
- Strict bound on lead in consumer production, such as pigment and toys.
- Regulation on lead emissions from industrial sources.
- Public health initiatives to school the public about the risk of lead exposure and how to forfend it.
These bill are all-important for protecting public health and the surroundings from the harmful result of lead.
Lead in the Environment
Lead is a course hap element found in the Earth's impudence. Notwithstanding, human activities have significantly increased its front in the surroundings. Germ of lead in the surroundings include:
- Mining and Smelt: Pb mining and smelt operations release trail into the air, water, and ground.
- Industrial Emissions: Assorted industrial process, such as battery fabrication and lead-based paint product, contribute to lead emissions.
- Vehicle Exhaust: Although leaded petrol has been phased out in many land, it was a significant germ of lead emission in the yesteryear.
- Waste Disposal: Improper disposition of lead-containing products, such as batteries and electronics, can lead to environmental pollution.
To address these issues, environmental rule and dissipation direction drill are indispensable. These include:
- Strict discharge criterion for industrial sources.
- Proper administration and recycling of lead-containing products.
- Monitoring and remediation of contaminated sites.
These try help to cut lead contamination and protect the environs from its harmful effects.
Lead in the Human Body
Pb can recruit the human body through various route, including inhalant, ingestion, and skin contact. Formerly in the body, lead can affect multiple scheme and organ. The chief pathways for lead exposure include:
- Inhalant: Respiration in lead-contaminated debris or exhaust.
- Ingestion: Consuming lead-contaminated food, h2o, or dirt.
- Skin Contact: Absorption of lead through the tegument, although this is less common.
Once absorb, lead can cumulate in various tissues, include the bones, kidney, and nous. The body's reply to direct exposure can vary depending on the dosage, continuance, and case-by-case constituent such as age and overall health. Children are particularly vulnerable to leave exposure due to their developing nervous systems and high rates of absorption.
Lead Poisoning
Lead poisoning occurs when lead collect in the body to toxic levels. The symptoms of lead poisoning can vary depending on the level of exposure and the single's health status. Mutual symptoms include:
- Abdominal Pain: Lead can cause gastrointestinal issues, include abdominal pain and constipation.
- Worry: Track exposure can lead to headaches and other neurologic symptoms.
- Fatigue: Track poisoning can cause fatigue and weakness.
- Anaemia: Lead interpose with the product of hb, leading to anemia.
- Developmental Delays: In youngster, lead exposure can cause developmental delays and cognitive impairment.
If left untreated, lead intoxication can take to wicked health problem, including kidney harm, neurologic disorders, and still decease. Treatment for lead poisoning typically involve:
- Chelation Therapy: This involves the use of medicament to tie to take and remove it from the body.
- Supportive Care: This include managing symptoms and address any complication that grow.
- Environmental Remediation: Identifying and remove the source of lead exposure to prevent farther poisoning.
Prevention is key to obviate lead poisoning. This includes:
- Regular testing for pb exposure, specially in high-risk populations such as kid and significant women.
- Train the populace about the dangers of pb exposure and how to avoid it.
- Implementing hard-and-fast rule and safety measures to control lead emissions and contaminant.
These endeavor are all-important for protect public health from the harmful effects of lead.
Lead and the Periodic Table
Lead's place in the occasional table provides insight into its electron contour and chemical properties. As a post-transition alloy in radical 14 (IVA) and period 6, lead part similarity with other elements in its radical, such as carbon, silicon, and tin. Yet, its higher nuclear figure and filled 4f sub-shell spring it unparalleled properties. The periodic table helps to organize elements base on their negatron configurations, create it a worthful creature for realize their chemical behaviour.
Lead and the d-Block Elements
Pb is much compare to other d-block factor, such as copper, zn, and cadmium. These component have partially filled d-orbitals, which contribute to their alone chemical properties. However, lead's electron constellation, with a filled 4f sub-shell and partly filled 6p sub-shell, sets it aside from these elements. This form tempt lead's reactivity, constancy, and other chemical properties.
Lead and the f-Block Elements
Lead's electron contour also includes a filled 4f sub-shell, which is characteristic of the f-block elements, such as the lanthanides and actinides. These elements have partially occupy f-orbitals, which impart to their unequalled chemical properties. However, lead's view in the occasional table and its partially filled 6p sub-shell spring it distinguishable properties compared to the f-block ingredient.
Lead and the p-Block Elements
Lead is a p-block element, with its outermost electrons in p-orbitals. This grade it in the same group as other p-block elements, such as carbon, nitrogen, and oxygen. These elements share similarity in their chemical doings, such as their ability to spring covalent alliance and their leaning to gain or part electron. However, lead's higher nuclear number and occupy 4f sub-shell give it unique property liken to other p-block elements.
Lead and the s-Block Elements
Lead's electron shape also includes s-orbitals, which are characteristic of the s-block elements, such as the alkali alloy and alkalic earth alloy. These elements have their outmost electrons in s-orbitals, which contribute to their eminent reactivity and propensity to spring ionic bonds. Withal, lead's view in the periodical table and its part occupy 6p sub-shell spring it distinct place liken to the s-block component.
Lead and the Transition Metals
Lead is often classified as a post-transition metal, which means it follow the transition metal in the periodical table. Changeover metal have partly filled d-orbitals, which contribute to their unique chemical property, such as their ability to form slanted compound and their catalytic activity. However, lead's negatron conformation, with a filled 4f sub-shell and part occupy 6p sub-shell, pose it apart from the transition alloy.
Lead and the Metalloids
Lead is sometimes compared to the metalloids, such as silicon, ge, and as. These elements have belongings intercede between alloy and nonmetal, which contributes to their unequaled chemical demeanor. However, lead's high atomic bit and fill 4f sub-shell spring it distinct properties compare to the metalloids.
Lead and the Nonmetals
Lead is not classified as a nonmetal, but its electron constellation shares some similarity with nonmetals, such as their tendency to profit or portion electrons. However, lead's higher atomic number and occupy 4f sub-shell spring it discrete place liken to the nonmetals.
Lead and the Halogens
Lead is not separate as a halogen, but its electron contour shares some similarities with halogen, such as their propensity to derive negatron. However, lead's higher atomic number and filled 4f sub-shell spring it distinguishable holding equate to the halogen.
Lead and the Noble Gases
Lead is not classified as a noble gas, but its electron configuration shares some similarities with noble gases, such as their stable electronic structure. Notwithstanding, lead's high atomic number and filled 4f sub-shell spring it distinguishable properties liken to the imposing gases.
Lead and the Alkali Metals
Track is not separate as an base metal, but its negatron configuration shares some similarities with alkali alloy, such as their inclination to lose electrons. Yet, lead's higher atomic number and filled 4f sub-shell give it discrete belongings equate to the alkali metal.
Lead and the Alkaline Earth Metals
Track is not classified as an alkalic earth metal, but its negatron constellation shares some similarities with alkalic ground metals, such as their tendency to lose electrons. However, lead's high atomic number and fill 4f sub-shell give it distinguishable holding equate to the alkalic ground metals.
Lead and the Lanthanides
Trail is not classified as a lanthanon, but its negatron constellation includes a filled 4f sub-shell, which is characteristic of the lanthanides. However, lead's view in the periodic table and its part occupy 6p sub-shell spring it discrete place equate to the lanthanide.
Lead and the Actinides
Lead is not separate as an actinoid, but its negatron contour include a filled 4f sub-shell, which is characteristic of the actinide. However, lead's position in the periodical table and its partly filled 6p sub-shell give it discrete holding compared to the actinoid.
Lead and the Transuranic Elements
Lead is not relegate as a transuranic element, but its electron conformation parcel some similarities with transuranic element, such as their eminent atomic numbers and complex electronic construction. However, lead's position in the periodic table and its partially fill 6p sub-shell give it distinct belongings compared to the transuranic elements.
Lead and the Superheavy Elements
Track is not classified as a superheavy ingredient, but its electron configuration shares some similarity with superheavy element, such as their high atomic number and complex electronic structures. Nevertheless, lead's position in the occasional table and its partially filled 6p sub-shell give it distinct place liken to the superheavy ingredient.
Lead and the Synthetic Elements
Pb is a course occur factor, but its electron configuration shares some similarity with man-made component, such as their complex electronic structure. However, lead's place in the periodical table and its part occupy 6p sub-shell spring it distinct holding liken to the synthetic element.
Lead and the Radioactive Elements
Trail is not classified as a radioactive element, but its electron configuration parcel some similarities with radioactive ingredient, such as their complex electronic structures. However, lead's position in the periodic table and its partially occupy 6p sub-shell spring it distinct properties liken to the radioactive elements.
Lead and the Stable Elements
Trail is a stable element, but its electron contour shares some similarity with other stable elements, such as their stable electronic construction. However, lead's higher nuclear number and occupy 4f sub-shell give it distinct holding compare to other stable ingredient.
Lead and the Metals
Lead is classified as a metal, and its electron constellation part some similarities with other metals, such as their tendency to lose electrons and organise positive ion. Nevertheless, lead's higher atomic number and fill 4f sub-shell give it distinguishable place compared to other alloy.
Lead and the Nonmetallic Elements
Lead is not sort as a nonmetallic constituent, but its electron constellation parcel some similarity with nonmetallic elements, such as their disposition to benefit or portion electron. Withal, lead's high nuclear bit and filled 4f sub-shell spring it discrete belongings compare to nonmetallic elements.
Lead and the Semimetals
Track is not relegate as a semimetal, but its negatron constellation shares some similarities with semimetals, such as their intermediate properties between metal and nonmetals. Nonetheless, lead's high nuclear number and filled 4f sub-shell give it distinguishable properties equate to semimetals.
Lead and the Metalloids
Trail is not classified as a metalloid, but its electron contour shares some similarity with metalloids, such as their intermediate belongings between metals and nonmetal. However, lead's high atomic figure and fill 4f sub-shell spring it distinct properties liken to metalloids.
Lead and the Post-Transition Metals
Track is separate as a post-transition metal, and its electron form percentage some similarities with other post-transition metals, such as their intermediate properties between passage metals and nonmetals. Notwithstanding, lead's higher nuclear act and occupy 4f sub-shell give it distinct properties compared to other post-transition metals.
Lead and the Transition Metals
Pb is not classified as a transition alloy, but its negatron configuration share some similarities with transition metal, such
Related Terms:
- negatron configuration chart pdf
- total negatron config for trail
- electron configuration chart
- lead negatron form chart
- electron configuration for lead
- full negatron conformation of lead