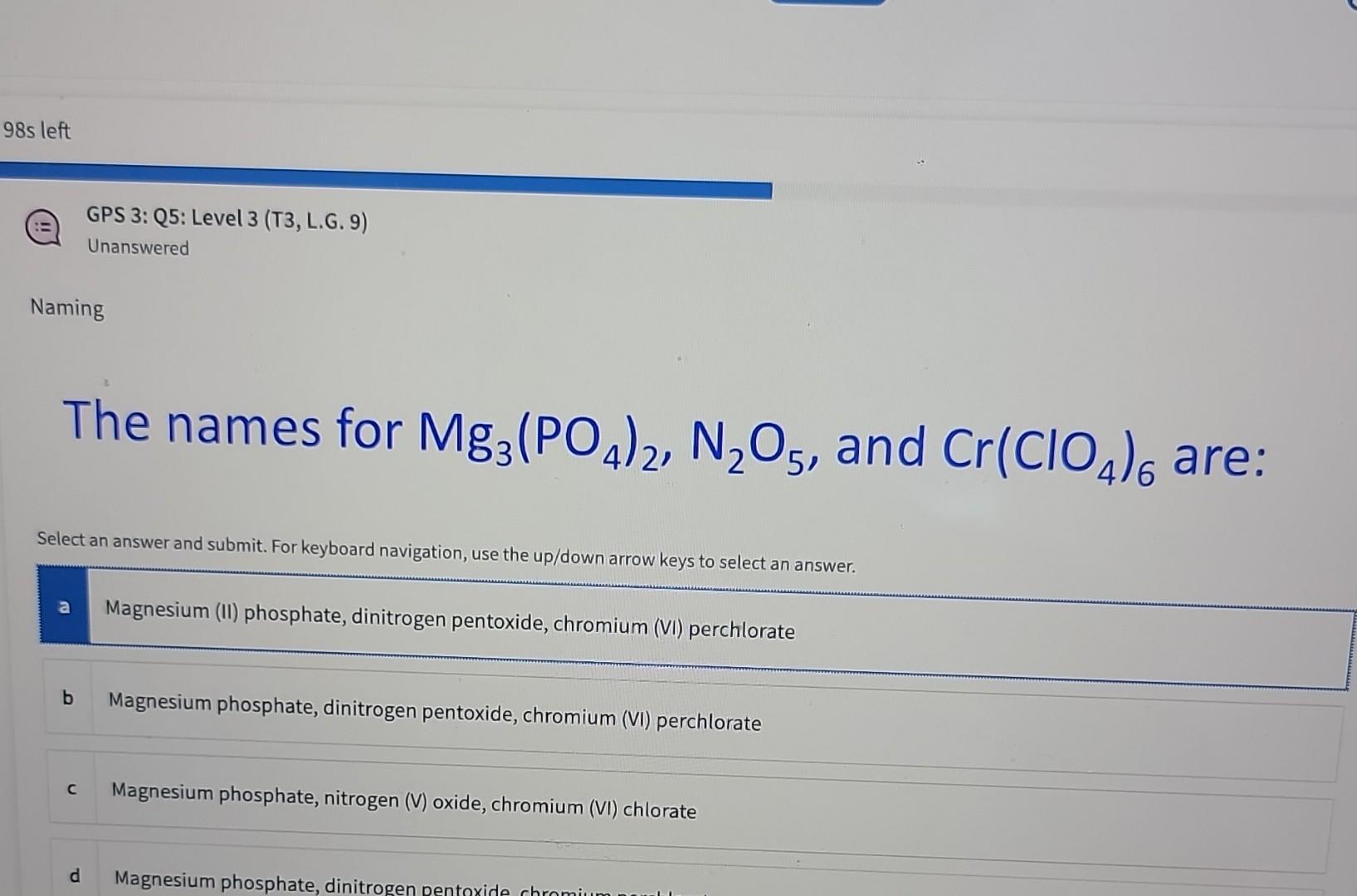
Magnesium phosphate, ofttimes denoted as Mg3 (PO4) 2, is a chemic compound that plays a crucial role in various industrial and biological processes. This compound is wide used in agriculture, medicine, and environmental applications due to its unequaled properties. Understanding the makeup, properties, and applications of Mg3 (PO4) 2 can provide worthful insights into its significance in different fields.
Composition and Properties of Mg3 (PO4) 2
Mg3 (PO4) 2 is composed of magnesium (Mg) and phosphate (PO4) ions. The chemic formula indicates that there are three magnesium ions for every two phosphate ions. This compound is typically found in a crystalline form and is known for its constancy and insolubility in h2o. The insolubility of Mg3 (PO4) 2 makes it an excellent choice for applications where a stable, non reactive substance is required.
Some of the key properties of Mg3 (PO4) 2 include:
- Chemical Formula: Mg3 (PO4) 2
- Molar Mass: 262. 86 g mol
- Appearance: White crystalline powder
- Solubility: Insoluble in water
- Melting Point: Decomposes before melting
Applications of Mg3 (PO4) 2
Mg3 (PO4) 2 has a blanket range of applications across several industries. Its unique properties make it a worthful compound in husbandry, medicine, and environmental science.
Agriculture
In agriculture, Mg3 (PO4) 2 is used as a fertilizer to ply essential nutrients to plants. Magnesium is a all-important element for plant growth, as it is involved in the synthesis of chlorophyll and the activation of enzymes. Phosphate, conversely, is essential for energy transference and storage in plants. By ply both magnesium and phosphate, Mg3 (PO4) 2 helps improve plant health and yield.
Some of the benefits of using Mg3 (PO4) 2 as a fertilizer include:
- Improved soil fertility
- Enhanced plant growth and yield
- Better nutrient uptake by plants
- Reduced need for disunite magnesium and phosphate fertilizers
Medicine
In the aesculapian field, Mg3 (PO4) 2 is used as a dietary supplement to treat magnesium deficiency. Magnesium is essential for various bodily functions, include muscle and nerve use, blood glucose control, and blood press rule. A deficiency in magnesium can direct to symptoms such as muscle cramps, fatigue, and irregular heartbeat. Mg3 (PO4) 2 supplements can help alleviate these symptoms by providing a pronto absorbable form of magnesium.
Some of the medical applications of Mg3 (PO4) 2 include:
- Treatment of magnesium deficiency
- Management of muscle cramps and spasms
- Support for cardiovascular health
- Improvement of bone health
Environmental Science
Mg3 (PO4) 2 is also used in environmental applications, particularly in h2o treatment and waste management. Its insolubility in water makes it an effective agent for withdraw phosphate from effluent, which helps prevent eutrophication in aquatic ecosystems. Eutrophication is a summons where excessive nutrients, such as phosphate, result to the overgrowth of algae and other aquatic plants, depleting oxygen levels and harm aquatic life.
Some of the environmental applications of Mg3 (PO4) 2 include:
- Phosphate removal from wastewater
- Prevention of eutrophication
- Soil remediation
- Waste management
Synthesis and Preparation of Mg3 (PO4) 2
Mg3 (PO4) 2 can be synthesized through diverse chemical reactions. One common method involves the reaction of magnesium hydroxide with phosphorous acid. The response can be represented as follows:
Note: The reaction should be carried out in a contain environment to ensure safety and purity of the product.
Mg (OH) 2 H3PO4 Mg3 (PO4) 2 H2O
Another method involves the reaction of magnesium carbonate with phosphorous acid:
Note: This method is oft preferred for its simplicity and the accessibility of reactants.
MgCO3 H3PO4 Mg3 (PO4) 2 CO2 H2O
The synthesis of Mg3 (PO4) 2 typically involves the follow steps:
- Preparation of Reactants: Ensure that the reactants are pure and in the correct stoichiometric ratios.
- Reaction: Mix the reactants in a worthy container and allow the reaction to proceed.
- Filtration: Filter the response concoction to disunite the solid Mg3 (PO4) 2 from the liquid.
- Drying: Dry the trickle Mg3 (PO4) 2 to remove any residual moisture.
- Purification: Purify the Mg3 (PO4) 2 if necessary, using techniques such as recrystallization or launder.
Safety and Handling of Mg3 (PO4) 2
While Mg3 (PO4) 2 is loosely regard safe, it is significant to handle it with care to avoid potential hazards. Some safety precautions to take include:
- Personal Protective Equipment (PPE): Wear reserve PPE, include gloves, safety glasses, and lab coats, when deal Mg3 (PO4) 2.
- Ventilation: Ensure adequate airing to prevent the accumulation of dust particles.
- Storage: Store Mg3 (PO4) 2 in a cool, dry place away from incompatible substances.
- Disposal: Dispose of Mg3 (PO4) 2 harmonize to local regulations and guidelines.
In case of accidental ingestion or inhalation, assay medical attending forthwith. While Mg3 (PO4) 2 is not extremely toxic, it can cause botheration to the skin, eyes, and respiratory tract.
Future Prospects of Mg3 (PO4) 2
The future of Mg3 (PO4) 2 looks promise, with likely applications in emerge fields such as nanotechnology and biomedicine. Researchers are research the use of Mg3 (PO4) 2 nanoparticles for targeted drug delivery and tissue organize. The unequaled properties of Mg3 (PO4) 2, such as its biocompatibility and biodegradability, make it an attractive material for these applications.
In gain, the grow demand for sustainable and environmentally friendly products is motor the development of new applications for Mg3 (PO4) 2. for example, Mg3 (PO4) 2 can be used as a component in eco friendly fertilizers and water treatment systems, helping to reduce the environmental impact of agriculture and industrial activities.
Some of the futurity prospects of Mg3 (PO4) 2 include:
- Nanotechnology: Development of Mg3 (PO4) 2 nanoparticles for drug delivery and tissue engineering
- Biomedicine: Use of Mg3 (PO4) 2 in biomedical applications, such as bone grafts and dental implants
- Environmental Science: Application of Mg3 (PO4) 2 in eco friendly fertilizers and h2o treatment systems
- Agriculture: Development of new Mg3 (PO4) 2 based fertilizers to improve crop yield and soil health
As inquiry continues to uncover new applications and benefits of Mg3 (PO4) 2, its importance in several industries is likely to turn. The versatility and unequaled properties of this compound create it a valuable imagination for direct current and future challenges in agriculture, medicine, and environmental science.
Mg3 (PO4) 2 is a versatile compound with a wide-eyed range of applications in farming, medicine, and environmental skill. Its unique properties, such as unsolvability in water and constancy, make it an idealistic choice for diverse industrial and biologic processes. As research continues to explore new applications and benefits of Mg3 (PO4) 2, its importance in different fields is likely to grow. Understanding the constitution, properties, and applications of Mg3 (PO4) 2 can render worthful insights into its signification and potential for futurity developments.
Related Terms:
- magnesium phosphate formula
- mg3 po4 2 ksp
- mg3 po4 2 mass
- mg3 po4 2 solubility
- mghpo4
- mg3 po4 2 formula